ISSN: 0970-938X (Print) | 0976-1683 (Electronic)
Biomedical Research
An International Journal of Medical Sciences
- Biomedical Research (2016) Volume 27, Issue 2
The effects of aqueous ginger extract on pancreas histology and on blood glucose in normal and alloxan monohydrate-induced diabetic rats.
| Maisa MA Al-Qudah1, Moawiya A Haddad2, Jafar MF EL-Qudah2* 1Department of Allied Medical Sciences, Zarqa University College, Al-Balqa Applied University, Al-Zarqa, Jordan 2Department of Nutrition and Food Processing, Faculty of Agricultural Technology, Al-Balqa Applied University, 19117 Al-Salt, Jordan |
| Corresponding Author: Jafar MF EL-Qudah, Faculty of Agricultural Technology, Department of Nutrition and Food Processing, Al-Balqa Applied University, Jordan |
| Accepted: January 20, 2016 |
This study aims to investigate the biochemical and histological changes in the pancreas of rats exposed to aqueous extract of ginger. Twenty adult female albino rats were divided into four groups of five each: Group I, the control group. Group II: Untreated diabetic rats (injected with 65mg/kg Body Weight (BW) of alloxan intraperitoneally); received distilled water. Group III: Diabetic rats; received 500 mg/kg BW/day of the ginger extract. Group IV: Diabetic rats; received 1000 mg/kg BW/day of the ginger extract. The treatment continued for 21 days, then rats were anesthetized, and their pancreases were extirpated to be processed for light microscopic examinations. The alloxan induced rats exhibited hyperglycemia accompanied with increases in glucose, Total Cholesterol (TC), Low Density Lipoprotein Cholesterol (LDL-C) and Triglycerides (TG) levels. On the other hand, there were reductions in insulin and High Density Lipoprotein Cholesterol (HDL-C) levels. Ginger was effective in lowering serum glucose and returned the other earlier mentioned blood assays levels in the ginger treated diabetic rats to almost normal value. Microscopic examination of the pancreatic sections of diabetic rats treated with ginger at either doses revealed approximately normal structure, compared with the untreated diabetic group. These results indicate that treatment with aqueous extract of ginger may be effective in lowering hyperglycemia and hyperlipidemia in diabetic rats.
Keywords |
||||||||||||
| Zingiberaceae, Light microscope, Lipid profile, Diabetes mellitus. | ||||||||||||
Introduction |
||||||||||||
| Pancreas is a vital endocrine-exocrine organ that produces several hormones and enzymes. Its enzymes help in the digestion of carbohydrates, fats, and proteins whereas its hormones such as insulin regulate carbohydrate metabolism in the body and maintains passage of glucose across the cell membrane [1]. Therefore, any change in the function of the organ may directly affect the physiological function of the body [2,3]. Diabetes mellitus is a chronic endocrine disease widely present in all parts of the world affecting nearly 10% of the population all over the world and considered as one of the leading cause of death in humans [4-6]. The World Health Organization (WHO) estimates that over 300 million people worldwide will have diabetes mellitus by the year 2025 [7]. Diabetes is characterized by multiple defects in its pathophysiology [8] and abnormalities in protein, fat and carbohydrate metabolism [9,10]. It is evident that this disease leads to hyperglycemia, and to many other complications such as hyperlipidemia, hypertension, atherosclerosis, retinopathy, neuropathy and nephropathy [5]. | ||||||||||||
| Zingiber officinal roscoe (Z. officinalee) is one of the Zingiberaceae family [11]. Z. officinalee is commonly called ginger. Ginger rhizome is widely used worldwide as a spice. It has been used as a medicine in Asian, Indian and Arabic herbal traditions [12]. Major active components of Z. officinale are thought to be volatile oils, and phenol compounds such as gingerols, Zingbrene and shogaols [13,14]. A lot of researches were done to clarify ginger's influence on lipid profile; HDLC, LDL-C, TC and Triglycerides (TG) [15]. Ginger's effect on glucose and lipid metabolism in both liver and blood was investigated [13,16]. It was reported that high dose (>6 g) intake of ginger might cause gastric reflux in human [17] and IgE-mediated allergy may be produced, due to ginger dust inhalation [18]. Thus, when ginger is consumed at a higher dosages and for long time, it could be toxic [19]. The present study aims to investigate the biochemical and histological changes in the pancreas of rats exposed to aqueous extract of ginger and to observe the hypoglycemic activity of ginger in alloxan induced diabetic female rats. | ||||||||||||
Materials and Methods |
||||||||||||
| The fresh rhizomes of ginger were obtained from a local market in Amman, Jordan. | ||||||||||||
| Preparation of plant extracts | ||||||||||||
| The raw extract was prepared according to the method used by Elshater et al., [20]. Plant parts were separated and washed with distilled water, dried and then grind using blender. Each 30 g powdered plant material was extracted by refluxing with 100 ml of hot water for two weeks at room temperature with shaking at 150 rpm. The extract was filtered through White canvas and filter paper. The mixture was filtered, then the filtrate was centrifuged and the clear supernatant fraction was separated. The concentration was considered to have 1g/ml based on the weight of the starting material. The extracts was purified by filtration through 0.22 μm filter units and kept at -20°C until use. | ||||||||||||
| Animals | ||||||||||||
| The study was performed on 20 healthy adult female albino rats, with a weight ranging from 190-210 g. The rats were obtained from the animal house of the Faculty of Medicine, University of Jordan, and kept under controlled laboratory conditions under a 12 h dark and 12 h light cycle, at 25°C, and were provided with a diet and water ad libitum. The study was approved by the Institutional Review Board of Al-Balqa Applied University and the University of Jordan, Amman. All procedures related to use of animals was conducted in compliance with ethical guidelines guaranteed by the intuition ethical committee. | ||||||||||||
| Induction of Diabetes | ||||||||||||
| Alloxan is known as a useful drug to induce diabetes in experimental animals [21]. After overnight fasting diabetes was induced in rats by intraperitoneal injection of alloxan monohydrate (Sigma, St. Louis, MO, USA) dissolved in 1ml distilled water at a dose of 65mg/kg BW [22]. After the injection, animals had free access of food and water, also after 6 hours of injection rats were given solution of 5% D-glucose solution for the next 24 h. Rats were allowed to stabilize for 3 days and blood samples were drawn from the eye vein on the 3rd day to determine the blood glucose concentrations to confirm the occurrence of diabetes, the rats which have blood glucose more than 150 mg/dl were considered diabetic and were used for further experiments [5,23,24]. The treatment with ginger extract was started after induction of diabetes by alloxan and considered as the first day of treatment. The treatment continued for 21 days. | ||||||||||||
| Experimental design | ||||||||||||
| The rats were randomly divided into 4 groups: I, II, III and IV, 5 animals in each group. Groups III and IV consisted of diabetic rats receiving daily an oral single dose of ginger plant extract through a stomach tube. All animals were fasted before the treatment. Group I (control group; did not receive alloxan), which included rats that were allowed water ad libitum and were fed a standard diet. Group II: Untreated diabetic rats (injected with 65 mg/kg BW of alloxan); received distilled water. Group III: Diabetic rats; received 500 mg/kg BW /day of the Ginger extract Group IV: Diabetic rats; received 1000 mg/kg BW /day of the Ginger extract. All rats were weighed before the start of the experiment and at the end of the experiment. | ||||||||||||
| At the end of the (21) days, the rats were sacrificed, relative pancreas weight was computed by expressing the absolute weight of the organ to the animal’s body weight as described below [25]. Relative organ weight=organ weight (g)/body weight (g) × 100. The pancreas was obtained and fixed in 10% Formalin solution for histological examination. It was trimmed, processed, embedded in paraffin wax, sectioned, placed on glass microscope slides, and stained with hematoxylin and eosin (H.E.). | ||||||||||||
| Collection of blood samples | ||||||||||||
| Blood samples were collected in clean plain tubes from eye vein of all animals three times and the serum was analyzed in laboratory to determine the serum levels of blood glucose and other biochemical parameters. The blood was collected firstly at the beginning of the experiment before injection of alloxan, then on the 3rd day after injection of alloxan to determine the blood glucose concentrations to confirm the development of diabetes mellitus and finally at the end of the experiment (three weeks after injection of alloxan). The collected blood was left to clot then centrifuged at 3500 rpm for 5 min. The serum was separated and stored at - 80°C for later analyses. | ||||||||||||
| Biochemical study | ||||||||||||
| Laboratory analyses were performed at Jabal Al-Hussein Laboratory, Amman, Jordan. The serum levels of blood glucose were measured using enzymatic colorimetric (GODPAP) method, according to the manufacturer’s instructions (BioSystem, Spain). Insulin was determined using the access ultrasensitive insulin assay (Beckman Coulter, Inc, California). Lipid assays (TG), (TC), (LDL-C) and (HDL-C) were performed using ACCENT-200 methods (CORMAY S.A., Poland). All tests were analyzed according to the manufacturer’s instructions. | ||||||||||||
| Statistical analyses | ||||||||||||
| Data for all groups were expressed as mean ± standard deviation. Statistical analyses were performed using one-way analysis of variance (ANOVA) and student t-test. Differences were considered to be statistically significant when P value was <0.05, <0.01 or <0.001. All statistical analyses were performed with SPSS software, (version 20 Inc. Chicago, Illinois, IL). | ||||||||||||
Results |
||||||||||||
| The final body weight and the percentage weight gain of the rats were not significantly (P>0.05) different from those of the control at the two doses of the extract investigated (Table 1). This study also showed that aqueous extract of ginger had no significant effect on body weight of both the diabetic and nondiabetic rats. Table 2 shows the effect of ginger on serum glucose concentration and insulin level in alloxan-diabetic rats. Administration of ginger to diabetic rats showed remarkably reduction in glucose concentration, there were very highly significant (P<0.001) in rats have 500 mg ginger/kg BW as well as 1000 mg ginger/kg BW when compared with group II (diabetic group). In diabetic rats there were a very highly significant (P<0.001) reduction in insulin level as compared with group I (control group). Concerning the rats treated with the two doses of ginger, there were elevation in insulin level but it was not significant (P>0.05) when compared with group II. Table 3 shows that alloxan produced significant increase in the levels of serum cholesterol (P<0.01), LDL-C (P<0.05), and TG (P<0.01), and a significant decrease in the levels of serum HDL-C (P<0.05), when compared with control group. Moreover, treatment with both doses of ginger extract to diabetic rats produced significant reduction in the levels of serum TC, when compared with diabetic group, while there was a significant (P<0.05) reduction in TG levels in rats treated with 1000 mg ginger/kg BW, when compared with diabetic group. | ||||||||||||
| The extract did not significantly (P>0.05) alter the level of HDL-C and LDL-C at both dose investigated. The computed pancreas body weight ratio of the diabetic rats and those treated with 500 mg ginger/kg BW, were not significantly (P>0.05) different from those of their control. | ||||||||||||
| However the computed pancreas body weight ratio significantly (P<0.05) increase in rats treated with 1000 mg ginger/kg BW, when compared with diabetic group (Table 4). | ||||||||||||
| Histopathological investigation | ||||||||||||
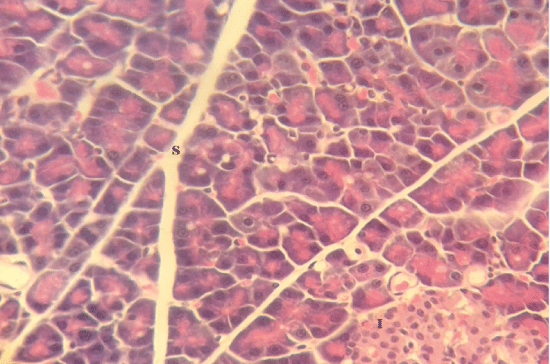
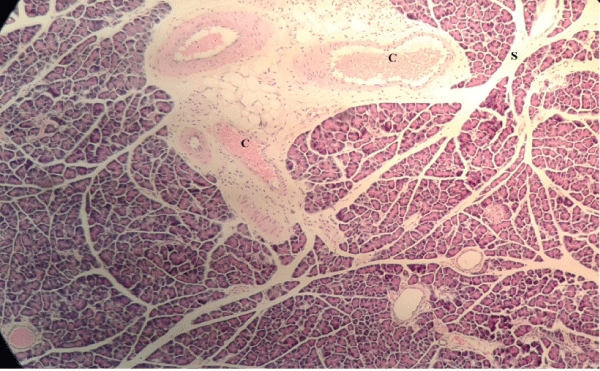
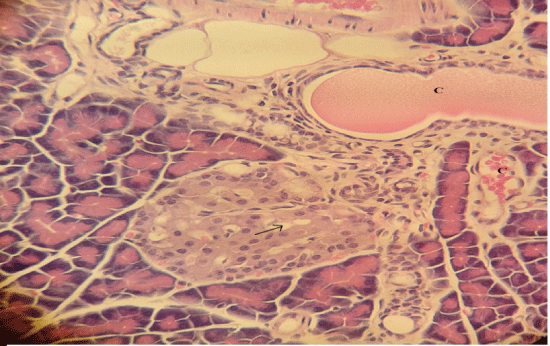
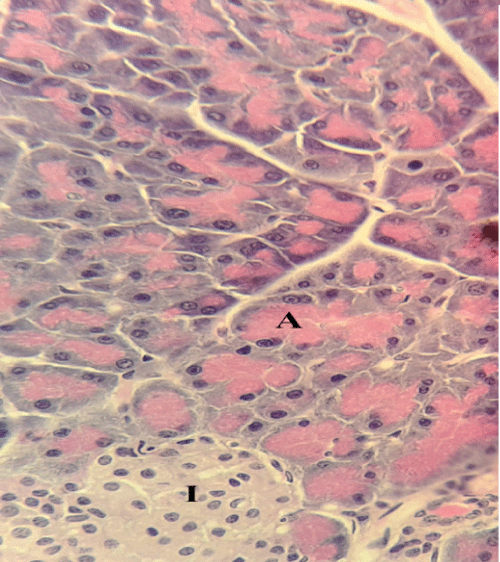
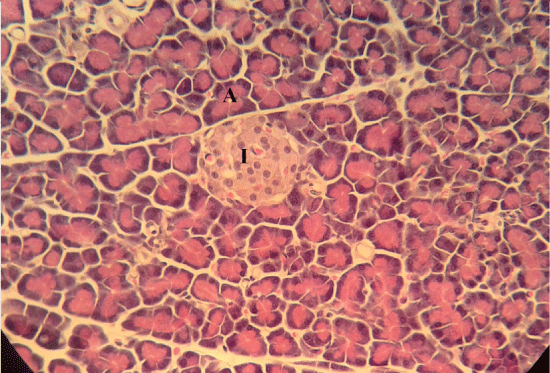
| Microscopically, pancreas from control rat group showed the normal histological structure, the histological appearance of the pancreatic islet cells showed no histopathological changes (Figure 1). In diabetic control rats, abnormalities in the pancreas were recorded. The congestion of RBCs in blood vessels (Figures 2 and 3) as well as thickening of the septa (Figure 2) were obviously seen. In addition, the islets of Langerhans have been prominently changed (Figures 3 and 4) and a slight vaculation of its cells was observed as shown in Figure 3. | ||||||||||||
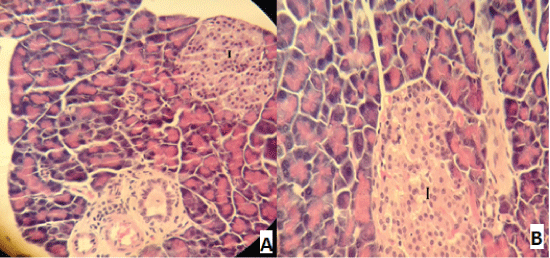
| Compared with the diabetic untreated group, approximately normal structure of acinar cells and islets of Langerhans were seen in animals given oral 500 mg/Kg BW of ginger (Figure 5). Moreover, Microscopic examination of the pancreatic sections of rats treated with 1000mg /Kg BW ginger revealed numerous small sized normal islets of Langerhans (Figure 6). | ||||||||||||
Discussion |
||||||||||||
| Diabetes is one of many leading causes of morbidity and mortality in all countries of the world. There are reports that several medicinal plant such as ginger rhizome have hypoglycemic and hypolipidemic effects [24,26]. The results of this study indicated that administration of ginger extract at 500 and 1000 mg/kg of diabetic female rats for 21 days decreased blood glucose level, TC and TG and increased serum insulin and HDL-C levels. These findings were in agreement with other previous studies [27,28]. Khattab et al., [26] found that diabetic rats exhibited significant improvement in biological evaluation, glucose, insulin and lipid profile, when compared with untreated diabetic group. | ||||||||||||
| The mechanism of hypolipedemic action of medicinal plant might be due to the inhibition of dietary lipid absorption in the intestine or its production by liver or stimulation of the bilary secretion of cholesterol and cholesterol excretion in the feces [29-31]. In this study, treatment of diabetic rats with aqueous ginger extract was significantly reduced serum glucose level in comparison with diabetic group. Although the reduction was significantly higher in comparison to normal control group, it was not enough to achieve normal values in control rats as illustrated in Table 2. These results are in agreement with previous studies [32]. | ||||||||||||
| In the current study the decreasing levels of serum triglycerides after the treatment at a dose of 1000 mg/kg BW with ginger extract may be due to the stimulating effect of ginger extract on insulin. A similar result was reported by Akhani et al., [33] who reported that treatment of diabetic rats with ginger produced a significant increase in insulin levels and a decrease in fasting glucose levels in diabetic rats. Treatment with ginger was also found to decrease in serum cholesterol, serum triglyceride and blood pressure in diabetic rats. Ozougwu and Eyo, [34] found that the increasing dosages (200, 250 and 300mg/kg BW) of Z. officinalee aqueous extracts produced dose-dependent significant (P<0.05) reductions in the blood glucose levels of diabetic rats. | ||||||||||||
| It has been reported that ginger interfere with the activities of some digestive enzymes in rats [19] such as pancreatic amylase and lipase [35]. Besides, in rabbit it acted on the liver to reduce cholesterol biosynthesis and may stimulate cholesterol’s conversion to bile acids and increase its fecal excretion [36]. In the current study, the results showed that, microscopic examination of the pancreatic sections of diabetic untreated group revealed necrosis and atrophy of β-cells of islets of Langerhans, and congestion of pancreatic blood vessels. This results are in accordance with the findings of other studies [26,37]. The number of normal islets of diabetic pancreas was reduced as recorded by Qinna and Badwan, [38]. As well as, the endochylema of islets reported to have serious necrosis and granular degeneration. | ||||||||||||
| The examined sections of pancreas for diabetic rats treated with both doses of ginger showed no histopathological changes. On the other hand, the sections for diabetic untreated rats revealed slight vacuolation of sporadic β-cells of islets of Langerhans. This result agree with other reports [39,40]. Ginger is a good source of antioxidant and therefore may be capable of preventing tissue damage, it can protect the pancreas tissues from lipid peroxidation on diabetic rats [41,42]. | ||||||||||||
| According to the literature, Streptozotocin and alloxan are considered as the most used diabetogenic compound for inducing diabetes in experimental animal models [38,43]. There is little risk of toxicity when used as a spice. Acute toxicity tests in mice found no mortality or adverse effect when ginger extract was given at doses up to 2.5 g/kg over a 7 day period. Increasing the dose to 3-3.5 g/kg leading to 10-35% mortality [15]. On the other hand, other researchers found that, daily doses administration of ginger 500, 1000 and 2000 mg/kg BW of female and male rats for 35 days, was not associated with any abnormalities and mortalities [19]. | ||||||||||||
Conclusion |
||||||||||||
| The levels of blood glucose, total serum lipids and total serum cholesterol, which were raised in alloxan induced diabetic rats can be lowered by ginger aqueous extracts. Histopathological investigation of pancreatic sections of diabetic rats treated with ginger at either doses, revealed approximately normal structure of acinar cells and islets of Langerhans, compared with the diabetic untreated group. Overall, it may be concluded that aqueous extract of ginger possesses hypoglycemic and hypolipidemic potential in alloxan induced diabetic rats. | ||||||||||||
Acknowledgment |
||||||||||||
| We are grateful to the University of Jordan, Amman for providing the research facilities. | ||||||||||||
Tables at a glance |
||||||||||||
|
||||||||||||
Figures at a glance |
||||||||||||
|
||||||||||||
References |
||||||||||||
|