ISSN: 0970-938X (Print) | 0976-1683 (Electronic)
Biomedical Research
An International Journal of Medical Sciences
Research Article - Biomedical Research (2017) Volume 28, Issue 21
The effect of 9-cis-retionic acid on expression of GRIM-1 in KM mouse bone cancer tissue
Xiang Bai1#, Yong Zhao2#, Yuan Zhang1#, Baoqing Yu1* and Jianming Huang1
1Department of Orthopedics, Shanghai Pudong Hospital, Fudan University Pudong Medical Center, 2800 Gongwei Road, Huinan Town, Pudong, Shanghai, China
2Department of Orthopedics, South Hospital of Shanghai Sixth People's Hospital, Jiaotong University, Shanghai, China
#These authors contributed equally to this work.
- *Corresponding Author:
- Baoqing Yu
Department of Orthopedics
Shanghai Pudong Hospital
Fudan University Pudong Medical Center, China
Accepted on September 08, 2017
Interferon/retinoic acid combined with induction of apoptosis related genes (genes associated with retinoid-IFN-induced mortality and GRIMs) is a new apoptosis-related gene family, which is a group of novel cell death regulators with IFN-β combined RA induced expression. In this study, the effects of 9- cis-retinoic acid (RA) on expression of gene associated with retinoid-interferon mortality-1 (GRIM-1) in KM mouse bone cancer tissue were investigated. The transplantable models of bone cancer in KM mice were established to study inhibitory effects of 9-cis-RA against tumor. Immunohistochemical technique was used to analyze the expression of GRIM-1 in KM mouse cancer tissue. The results showed that 9- cis-RA was able to inhibit the growth of bone cancer on mouse at a dose of 650 mg/kg. The expression of GRIM-1 was declined in the mice treated with 9-cis-RA. 9-cis-RA has a significant antitumor effect in vitro. The mechanisms of 9-cis-RA on tumor might associate with down regulating expression of GRIM-1. There was no significant change in the transcriptional level of GRIM-1 in control bone tissue after 9-cis-RA treatment for 24 h. The transcription level of GRIM-1 in bone cancer tissues significantly increased.
Keywords
Bone cancer, Retionic acid (RA), Gene associated with retinoid-interferon mortality-1 (GRIM-1), Immumohistochemical, KM mouse, Gene expression.
Introduction
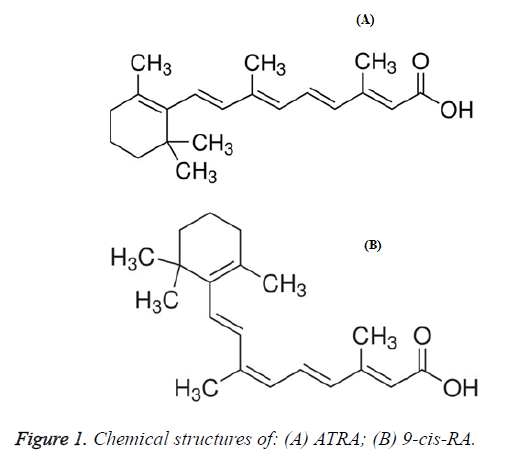
Retinoic acid (RA) is a natural or synthetic derivative from vitamin A, the metabolites of vitamin A having very important physiological activities exist in human body include all-trans-retinoic acid (ATRA), 9-cis-retinoic acid (9-cis-RA) etc. [1-4]. The chemical structure of 9-cis-RA is shown in Figure 1.
Lack of RA usually results in bronchial squamous epithelium growth and dys-differentiation, cellular fission and nucleic acid synthesis associated with increased mucosal metaplasia, and generates changes in pathological morphology which are similar to precancerous lesion [2-4]. Administration of RA can reverse the abnormal differentiation of epithelium and suppress the formation of chemicals-induced bronchial tumor. 9-cis-RA is able to induce differentiation of tumor cells and consequently promote apoptosis of tumor cells [1,3,4].
Bone cancer is a very common malignant tumor all over the world, nowadays it has become the primary cause of cancer death in most countries. Therefore bone cancer is currently considered to be the most serious malignant tumor to threaten health and life of human. When patients were diagnosed as suffering from bone cancer, approximately 70% of the cases belong to middle or terminal cancer. Early detection of bone cancer will significantly increase the 5-y survival rate of patients [5-15]. Multiple genetic abnormalities can be detected during the stages of early bone cancer or precancerous lesions, and these abnormalities usually precede clinical symptoms to become a molecular marker for early bone cancer to a certain extent. Interferon/retinoic acid combined with induction of apoptosis related genes (genes associated with retinoid-IFN-induced mortality and GRIMs) is a new apoptosis-related gene family, which is a group of novel cell death regulators with IFN-β combined RA induced expression. GRIM was found in 1994 with separation using antisense gene knockout method [16]. Retinoic acid/interferon induced apoptosis related gene 1 (GRIM-1) is a newly discovered cell apoptosis regulating gene, it is a new protein encoding a molecular weight of 64 kD, participate in interferon and retinoic acid induced apoptosis of tumor cell regulation [14]. The allelic gene SHQ-1 of GRIM-1 is an important part of the snoRNA, SHQ-1 was first discovered in the analogues of human as assembly factor shq1p of yeast H/ACA RNP, and it has very important functions in the biosynthesis of the H/ACA nucleoli and telomerase RNPs.
While there has been little to no report published on the expression level of the GRIM-1 protein in bone cancer tissues [17-21].
In this paper, polyphenolic compounds were used as quality control index and to observe the effect of 9-cis-RA on growth of Bone cancer in KM mice. Reverse transcription polymerase chain reaction (RT-PCR) was used to detect mRNA levels in tissues of bone cancer to explore the possible mechanisms of the anti-tumor effects of RA. This study provides a theoretical fundamental for the clinical application of RA in the treatment of bone cancer.
Materials and Methods
Experimental animals and reagents
9-cis-RA (HPLC grade) was purchased from Sigma, the chemical structure is shown in Figure 1. KM mice were provided by Shanghai Laboratory Animal Research Center, grade: clean level mice, age: 6-8 weeks, weight: 20 ± 2. The numbers of male and female are equal; the mice are divided into two groups according to sex. Bone cancer tumor was provided by the Chinese Academy of Sciences, Tianjin Institute of Materia Medica. RT-PRC reagent kits (Instant PCR Kit 3.0) and total DNA extraction reagent (Tiandz RNAiso Reagent) were purchased from Beijing Tiandz, Inc. PCR maker was purchased from Beijing Tianyu Biology Company (200-1500 bp).
Instruments
Olympus inverted microscope (Japan Olympus products); High precision electronic balance machine (Beijing Kerui); TLL-C centrifuge (Academy of Military Medical Sciences experimental instrument factory products); TGL-168 high-speed centrifuge (Shanghai Anting Scientific Instrument Factory products) UV-120-02 UV-visible spectrophotometer: (Shimadzu Corporation products); MiniCyclerTM amplification instrument (the Perkin Elmer (PE) products); DY-600 type medium voltage electrophoresis apparatus (the Becton Dickinson (BD) in products); ZF-1 UV transilluminator (Shanghai Changming optical electronic instrument factory products); Smart View gel scanning analyzer (Shanghai Furi biological experiment technology institute products).
Anti-tumor experiment
Bone cancer was stripped from tumor-bearing KM mice under sterile condition, well-growth tumor blocks were selected, and cut into small pieces by scissors, the cancer tissue blocks with similar size (approximately 2 × 124 Bone cancer cells) were used and transplanted to right axillary subcutaneous of mice by trocar, the mice were randomly divided into 6 groups, after inoculation for 24 h administration started every day and continued for 7 d. According to various 9-cis-RA dosage three 9-cis-RA groups (450 mg/kg, 650 mg/kg and 850 mg/kg per day) were set as well as a saline group as negative control group and cyclophosphamide positive control group (50 mg/kg per day by oral administration). The mice were divided into two groups to conduct experiments, after 24 h of final administration, the mice were executed by cervical dislocation method, weighed, dissected to strip the cancer block, and weighed the tumor. Tumor inhibition rate was calculated according to Eq 1.
Tumor inhibition rate=1-(average tumor weight of treatment group/average tumor weight of control group) × 100%----(1)
Homogeneity of variance test was first conducted with statistics processing method followed by t-test, P<0.05 indicates that the results are statistically significant.
Immunohistochemical staining
Samples were taken from Lewis cancer tissues of the second batch of mice after 7 d of 9-cis-RA administration. The samples of negative control group and positive control group were also taken. The procedures of immunohistochemistry were described as follows: the specimens were fixed with 10% neutral formalin, cut sections with paraffin embedded method. All the paraffin wax blocks were cut sections continuously, the thickness was about 4 μm. After conventional de-waxing, alcohol dehydration, and antigen repair with microwave oven, the samples were detected by SP immunohistochemical assay. The concentration of GRIM-1 was set as standard. A blank control with known GRIM-1 positive staining of bone cancer tissue sections was used as positive control.
Results
The effects of 9-cis-RA on the growth of transplanted tumor bone cancer
The first batch of experiments indicates that 9-cis-RA can inhibit the growth of primary tumor of bone cancer at dosage of 650 mg/kg and 850 mg/kg, with the tumor inhibition rate of 24.7% and 32.6% respectively. P<0.05 compared with the normal saline control group. The second batch of experiments shows that 9-cis-RA at dosage of 650 mg/kg and 850 mg/kg can inhibit the growth of primary tumor of bone cancer in KM mice, with the tumor inhibition rate of 36.1%, 40.2%, respectively. P<0.05 compared with the normal saline control group. The data were listed in Tables 1 and 2.
| Group | Dosage (mg/kg) | n (start/end) | Tumor weight (g) | Tumor inhibition rate (%) |
|---|---|---|---|---|
| Control group | - | 16/16 | 1.36 ± 0.82 | - |
| 9-cis-RA | 450 | 43081 | 1.12 ± 0.67 | 15.8 |
| 650 | 43081 | 0.87 ± 0.46 | 24.7 | |
| 850 | 43081 | 0.65 ± 0.31 | 32.6 |
Table 1. Effect of 9-cis-RA on tumor growth in KM mice bearing bone cancer (the first batch).
| Group | Dosage (mg/kg) | n (start/end) | Tumor weight (g) | Tumor inhibition rate (%) |
|---|---|---|---|---|
| Control group | - | 16/16 | 1.33 ± 0.78 | - |
| 9-cis-RA | 450 | 43081 | 1.09 ± 0.59 | 28.3 |
| 650 | 43081 | 0.82 ± 0.45 | 36.1 | |
| 850 | 43081 | 0.64 ± 0.29 | 40.2 |
Table 2. Effect of 9-cis-RA on tumor growth in KM mice bearing bone cancer (the second batch).
Effect of 9-cis-RA on the expression of GRIM-1
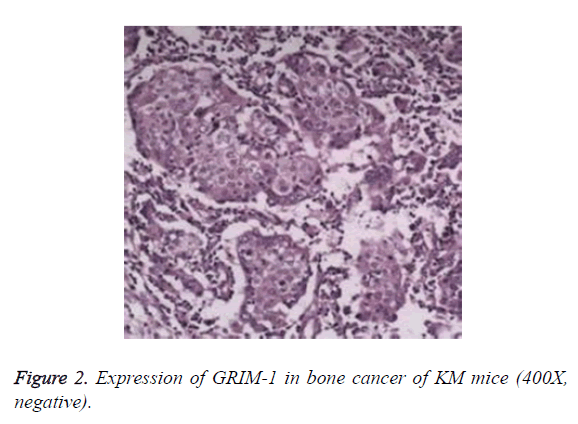
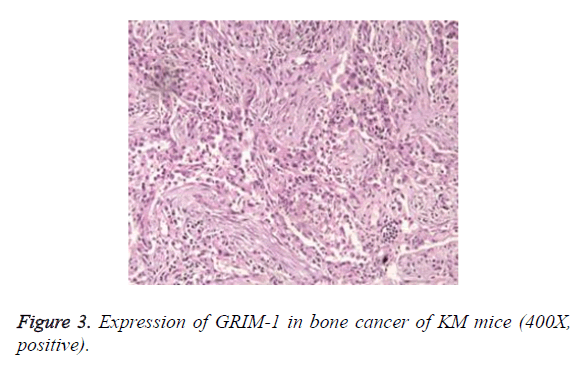
The staining results showed that no expression of GRIM-1 was observed in normal epithelial tissue. In the tissues of bone cancer, GRIM-1 had a higher rate of positive expression (Figures 2 and 3). After 7 d of different 9-cis-RA concentrations in KM mice, GRIM-1 positive expression rates were 53.2%, 44.1%, and 42.7%, respectively. The groups of 9- cis-RA dosage-650 mg/kg and 850 mg/kg showed statistically significant (P<0.05) comparing the normal saline control group. However, there were no significant changes (P>0.05) for cyclophosphamide control group of GRIM-1 positive expression rate.
Discussion
During the past two decades, RA compounds have received increasing attention for the prevention and treatment of solid tumor metastasis. Both clinical experiments and clinical practice indicate that RA has the function of inhibiting tumor metastasis and proliferation. ATRA induced differentiation has good effect in the treatment of acute promyelocytic leukemia. In recent years, it was found that RA also have an effect on growth of breast cancer and prostate cancer cells, RA is currently used in phase II clinical experiments for treatment of bone cancer. 9-cis-RA is a metabolite of ATRA in vivo, the metabolism is more effective in the process of induced cell differentiation with less adverse reactions like ATRA.
9-cis-RA can promote heterodimeric reaction of receptor, thus it has more effective functions on inhibiting tumor cell growth, promoting tumor cell differentiation and apoptosis. The ability of 9-cis-RA to induce apoptosis of tumor cells and decrease apoptosis gene protein expression are significantly stronger than ATRA. Apoptosis is extremely important for maintenance of tissue homeostasis. In recent years, with an in-depth investigation of apoptosis, scientists have found that apoptosis is closely related to tumor. Therefore, it becomes one of the new strategies for clinical treatment and prevention of cancer.
Previous study has found that GRIM-1 mainly regulated cell differentiation and/or apoptosis, the gene loss or expression abnormalities of GRIM-1 might have a certain relationship with carcinogenesisis, it is a potential gene for tumor suppressor [5-17]. Decrease or deficiency in the level of GRIM-1 gene transcription can enable epithelial cells to escape from the control of natural RA. In previous studies, 9-cis-RA was used for human bone adenocarcinoma strains of Calu-3 and H1975, human squamous cell carcinoma strains of H520 and CHLH-1, respectively [6,8-12,15]. The results indicated that 9-cis-RA can obviously inhibit bone cancer cell growth and promote cell apoptosis. RT-PCR detection showed that GRIM-1 transcription levels significantly increased, suggesting that GRIM-1 may involve in and mediate the process of tumor growth inhibition by 9-cis-RA. Because of the difference between many biological properties of cell strains and in vivo tumor cells, the primary cultured bone cancer cells were used for the current research to get closer to the environment in vivo. The bone cancer tissue and control bone tissue after treated with 9-cis-RA were observed and compared for GRIM-1 transcriptional level changes to explore the relationship between abnormalities of RA receptor expression and carcinogenesisis of bone cancer.
Singh et al. have found that the expression of GRIM-1 decreased in around 75% of cell lines, no expression of GRIM-1 was found for the undifferentiated bronchial epithelial cells cultured in vitro and the majority of pulmonary adenocarcinoma cells, but GRIM-1 was expressed in all normal bone tissues [19,20]. The results in this study showed that before 9-cis-RA treatment, the expression level of GRIM-1 in bone cancer tissue decreased compared with normal control group. Therefore it can be inferred that GRIM-1 may be significant to trigger differentiation and restriction of cell division (normal tissue), and a decrease of its expression might directly result in pathogenesis of bone cancer.
In the study of bone cancer cell line A549, it was found that after the bone cancer cells transfected by GRIM-1 with RA, both the growth rate of cells in vitro and rate of oncogenicity in vivo decreased [6,16-18]. The current results showed that there was no significant change in the transcriptional level of GRIM-1 in control bone tissue after 9-cis-RA treatment for 24 h.
Conclusion
The transcriptional level of GRIM-1 in bone cancer tissues significantly increased. Furthermore, the increased transcriptional level and the basic transcriptional level of control bone tissue are similar. There was no statistical significance of GRIM-1 transcriptional differences between bone cancer tissue, suggesting that RA can induce the increase of GRIM-1 and RA might be related to inhibition of tumor growth.
Acknowledgement
This work is funded by research project (No. 20134391) of Shanghai Municipal Health and Family Planning Commission.
References
- Duester G. Retinoic acid synthesis and signaling during early organogenesis. Cell 2008; 134: 921-931.
- Holland LZ. Developmental biology: A chordate with a difference. Nature 2007; 447: 153-155.
- Venkatesh K, Srikanth L, Vengamma B, Chandrasekhar C, Sanjeevkumar A, Mouleshwara Prasad BC, Sarma PV. In vitro differentiation of cultured human CD34+ cells into astrocytes. Neurol India 2013; 61: 383-388.
- Vanpelt HMM, Derooij DG. Retinoic acid is able to reinitiate spermatogenesis in vitamin A-deficient rats and high replicate doses support the full development of spermatogenic cells. Endocrinol 1991; 128: 697-704.
- Medhurst SJ, Walker K, Bowes M, Kidd BL, Glatt M, Muller M. A rat model of bone cancer pain. Pain 2002; 96: 129-140.
- Goss JR, Harley CF, Marina Mata MD, O'Malley ME, Goins WF, Hu X. Herpes vector-mediated expression of pro-enkephalin reduces bone cancer pain. Annals Neurol 2002; 52: 662-665.
- Hai L, Ben L, Yu Q, Zhang. Modulation of na_v1.8 by lysophosphatidic acid in the induction of bone cancer pain. Int J Precision Eng Manufacturing 2006; 14: 445-454.
- Li Q, Zhang X. Epigallocatechin-3-gallate attenuates bone cancer pain involving decreasing spinal tumor necrosis factor-α expression in a mouse model. Int Immunopharmacol 2015; 29: 818-823.
- Yang Y, Li H, Li TT, Luo H, Gu XY, Lü N. Delayed activation of spinal microglia contributes to the maintenance of bone cancer pain in female wistar rats via p2x7 receptor and il-18. J Neurosci 2015; 35: 7950-7963.
- Miles F, Molligan J, Ogunnaike B, Golen KV, Sikes R, Cooper C. Tgf-β1 regulation of prostate cancer cell adhesion to bone marrow endothelium. Malaria J 2015; 14: 1-7.
- Preston G, Meiklejohn D, Thomas N. Leucoerythroblastic anaemia, abnormal bone scan and prostate cancer with a primary haematological diagnosis. Brit J Haematol 2015; 168: 161.
- Reagan MR, Rosen CJ. Navigating the bone marrow niche: translational insights and cancer-driven dysfunction. Nature Rev Rheumatol 2016; 12: 154.
- Seki Y, Wakaki K. Pathological findings in a case of bone marrow carcinosis due to gastric cancer complicated by disseminated intravascular coagulation and thrombotic microangiopathy. Int J Hematol 2016; 104: 506-511.
- Bjelic-Radisic V, Stöger H, Winter R, Beham-Schmid C, Petru E. Long-term control of bone marrow carcinosis and severe thrombocytopenia with standard-dose chemotherapy in a breast cancer patient: a case report. Anticancer Res 2006; 26: 1627-1630.
- Hayashi S, Suzuki M, Kaoru TS, Izuha A, Noguchi C, Murakawa M. Usefulness of percutaneous injection of bone cement into calcaneotibial bone with cancer metastases for pain relief. J Japan Society Pain Clin 2009; 14: 19-22.
- Ménard R, Sansonetti P, Parsot C. Beaumont studies use of bone cement for spinal cancer. Embo J 1994; 13: 5293-5302.
- Lim YK, Kwak JW, Kim DW, Shin DH, Yoon MG, Ahn SH. Su-ff-j-71: a prototype fiducial marker composed of gold nanoparticles and bone cement for proton therapy of prostate cancers. Med Physics 2009; 36: 2492.
- Mccaffrey G, Thompson ML, Majuta L, Fealk MN, Chartier S, Longo G. Ngf blockade at early times during bone cancer development attenuates bone destruction and increases limb use. Cancer Res 2014; 74: 7014.
- Singh RK, Srivastava M, Prasad NK, Shetty PH, Kannan S. Hyperthermia effect and antibacterial efficacy of fe(3+) /co(2+) co-substitutions in β-ca3 (po4 )2 for bone cancer and defect therapy. J Biomed Mater Res B Appl Biomater 2017; 3: 1-12.
- Lin PP, Kang HG, Kim YI, Kim JH, Han SK. Minimally invasive surgery for femoral neck fractures using bone cement infusible hollow-perforated screw in high-risk patients with advanced cancer. Surg Oncol 2015; 24: 226.
- Carlsson E, Mestres G, Treerattrakoon K, López A, Karlsson OM, Larsson S. In vitro and in vivo response to low-modulus pmma-based bone cement. Biomed Res Int 2015; 1: 1-9.