ISSN: 0970-938X (Print) | 0976-1683 (Electronic)
Biomedical Research
An International Journal of Medical Sciences
Research Article - Biomedical Research (2017) Volume 28, Issue 11
Roles of pulmonary vascular endothelial cell injury and dimethylargininedimethylaminohydrolase/nitric oxide synthase/nitric oxide-system changes in the occurrence of diabetic sepsis in rats
Xiujuan Liu1, Fuzai Yin1, Kun Wei1, Yue Zheng1, Lanxiang Liu1, Fang Qiu1, Yonghong Xie1, Shufeng Xu1, En Mu2, Yingjian Liang2, Zhidan Zhang2 and Xiaochun Ma2*
1Department of Critical Care Medicine, First Hospital of Qinhuangdao, Qinhuangdao, PR China
2Department of Critical Care Medicine, the First Affiliated Hospital of China Medical University, Shenyang, PR China
- *Corresponding Author:
- Xiaochun Ma
Department of Critical Care Medicine
The First Affiliated Hospital of China Medical University, PR China
Accepted date: March 24, 2017
The aim of this study was to investigate the roles of Pulmonary Microvascular Endothelial Cell Injury (PMECI) and Nitric Oxide (NO) system in diabetic sepsis in rats. 64 Wistar rats were randomly divided into group A (the normal control group), group B (the sepsis group), group C (the diabetes group), and group D (the diabetic sepsis group), the content of pulmonary Evans blue (EBD) was measured; the pulmonary microvascular permeability was observed; specific Tyrosine kinase receptor 2 (Tie-2) mRNA in whole blood was measured by Polymerase Chain Reaction (PCR) assay to understand the amount of endothelial cells in the circulation; the content of NO in serum/lung tissues, as well as the content of inducible Nitric Oxide Synthase (iNOS) mRNA/endothelial Nitric Oxide Synthase (eNOS) mRNA in lung tissues, were detected to observe the roles of NO system. The EBD content in group D was the highest, followed by group B, and exhibited statistical difference than group A and C (P<0.01); Tie-2 mRNA in group D was significantly increased than the other three groups (P<0.01); the NO contents in the serum and lung tissues of group B/D were higher than the other two groups. The iNOS mRNA content in group D was the highest, while that of eNOS mRNA was the lowest, and the differences were statistically significant (P<0.05). Diabetes associated with sepsis might aggravate endothelial cell injury, and it was considered to be related with the imbalanced adjustments of the NO system.
Keywords
Diabetes, Sepsis, Pulmonary microvascular endothelial cell, Nitric oxide.
Introduction
Diabetes is prone to cause infections, especially chronic, lowgrade inflammation [1], and the probability of sepsis is relatively higher [2]. Take acute pancreatitis for example, it has been demonstrated that diabetes could increases parameters of systemic inflammation during the process [3]. Nevertheless, the findings of clinical large-sample epidemiological investigations about the impacts of diabetes on sepsis are not consistent, certain studies suggested that diabetes could increase the mortality of sepsis [4], while Vincent et al. [5,6] pointed out that diabetes had no effect on the mortality of sepsis. A large experimental study of sepsis [7] found that diabetes was not easy to develop into respiratory failure, and another 383,238-case large-sample study [8] even suggested that diabetes had protective effects on acute lung injury. Therefore, the impacts of diabetes on sepsis had become the controversial issue to be resolved.
Sepsis is a systemic inflammatory response caused by various pathogenic microorganisms or their toxins present inside blood or tissues, which is also named as Systemic Inflammatory Response Syndrome (SIRS) [9]. Currently, it has been considered that sepsis is not only caused by the infections, but the state of body's response to the infections should also be importantly emphasized [10]. Diabetes exhibited the suppressive status towards the infection-caused inflammatory response, and it seemed not to support the body’s excessive activation status when diabetic sepsis occurred, diabetes is pathologically characterized by microvascular disease, and the excessive activation of microvascular endothelial cells would be involved in the pathogenesis of sepsis. We speculated that, when the infective factors stimulated, the endothelial cells in diabetic body might appear different changes, and were involved in the occurrence of sepsis. This is very dangerous since it has been found that sepsis is the single most common factor causing Acute Renal Failure (ARF) in diabetes [11]. NO is one of the most important media that could maintain the functions of vascular endothelial cells, NO has the roles like “double-edged sword” inside the body with sepsis, too much or too little synthesis of NO would injure the body. NO is transformed from L-arginine (L-arg) under the catalysis of nitric oxide synthase. Recent studies had found that there existed endogenous nitric oxide synthase (NOS) inhibitor in vivo, namely Asymmetric Dimethylarginine (ADMA), which could competitively inhibit NOS’s activities, and reduce the generation of NO. ADMA is the competitive inhibitor of all three-configuration NOS. DDAH could degrade ADMA, the disorder of NO synthesis would cause the imbalance of endothelial cells’ homeostasis, thus involving the pathogeneses of many diseases. This study observed the changes of NO regulation system in diabetic sepsis, aiming to investigate the impacts on the functions of endothelial cells.
Materials and Methods
Animal grouping and model preparation
64 Wistar rats were randomly divided into 4 groups, namely the normal control group (group A, n=16), the diabetes group (group B, n=16), the sepsis group (group C, n=16), and the diabetic sepsis group (group D, n=16). Streptozotocin (STZ, sigma, USA) was intraperitoneally injected once (60 mg/kg body weight), group A was injected with the same amount of citric acid-sodium citrate buffer solution. And the random blood glucose ≥ 16.67 mmol/l within 48 h meant the success of preparing the diabetes model. After raised for four weeks, the Week 4 group and group A were fasted for 12 h, then intraperitoneally injected E. coli lipoprotein polysaccharide (LPS, sigma, USA), 10 mg/kg, and sampled 12 h later. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of China Medical University.
Sample preparation
After successfully anesthetized the animal, the abdominal artery blood was sampled and the right lung lower lobe was cut and stored at -70°C for RNA extraction. Rats were killed, take the left lung conventional paraformaldehyde fixation solution for 1 week, then dehydrated, embedded in paraffin, sliced and HE staining. Total RNA was extracted from blood and lung tissue using RNA simple Total RNA Kit (Tiangen, Beijing, China) according to the manufacturer’s instruction. The concentration and purity of the RNA in each sample were determined using a spectrophotometer. Reverse transcription was performed on 1 μg of total RNA from each sample using Super M-MLV (BioTeke, Beijing, China) and oligo (dT) 15. Then the mRNA levels of tie-2, eNOS, iNOS and DDAH2 were measured using RT-PCR (SYBR GREEN method) with cDNA as template and primers. Mouse GAPDH was served as an internal control. The relative mRNA level was calculated using 2-ΔΔCT method [12]. Primers for quantitative real time PCR, Primer sequences (TAKARA Co., Japan).
DDAH2-F: 5’-CAGATGACGCAGCGAGTG-3’;
DDAH2-R: 5’-CCAGTTCCGAGCAGGACA-3’;
iNOS-F: 5’-TCACCTATCGCACCCG-3’;
iNOS-R: 5’-CACTCCGCACAAAGCAG-3’;
eNOS-F: 5’-CTGGCAAGACAGACTACACGA-3’;
eNOS-R: 5’-CATCGCCGCAGACAAAC-3’;
GAPDH-F: 5’-TGTGTCCGTCGTGGATCTGA-3’;
GAPDH-R: 5’-TTGCTGTTGAAGTCGCAGGAG-3’.
Determination of Evans blue permeability
2% Evans blue (EBD, 20 mg/kg, sigma, USA) was injected through tail vein, 1h later, used saline to wash pulmonary vascular bed, cut the right lung lower lobe, weighed, added formamide and performed homogenization, then incubated at 37°C for 18 h and centrifuged at 12000 rpm for 20 minutes, the supernatant was then taken for measuring the OD value at 620 nm, the EBD content was then determined according to the standard curve.
Statistical analysis
The results were presented as means ± SD. Differences between groups were conducted using a one-way Analysis of Variance (ANOVA) and Bonferroni’s Multiple Comparison. The processing of the data and figures was performed using SPSS15.0 software. P<0.05 was considered to be significant.
Results
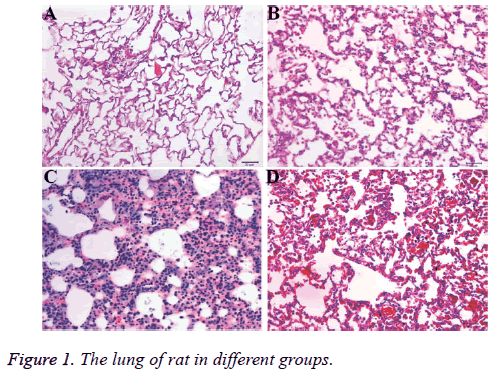
Pathological section with HE staining
Compared with Group A, the alveolar walls were thickened with pulmonary interstitial hyperplasia in Group B, as well as angiogenesis and inflammatory cell infiltration, including macrophages, plasmocyte and lymphocytes (Figures 1A and 1B); Group C mainly assumed lung consolidation and effusion, alveolar septum, infiltration with macrophages/eosinophils/ neutrophils, is thickened, with telangiectasia and punctate hemorrhage (Figure 1C). While the inflammatory reaction in Group D was remitted than Group B with a few macrophages, which was closed to alveolar and organizational structure of the normal alveolar septa with slightly thickened (Figure 1D).
EBD contents in lung tissue
The EBD content in group D was the highest (3.76 ± 0.77 mg/ ml), and equivalent to 2-fold of that in group B, while the contents of EBD in group A and C were both <1 mg/ml, there was statistically significant difference between group B and A (P<0.01), and there was significant difference between group D and the other three groups (P<0.01, Table 1).
| Group | Rats (n) | EBD content in lung tissues (mg/ml) |
|---|---|---|
| A | 5 | 0.70±0.21 |
| B | 5 | 1.74±0.24** |
| C | 5 | 0.93±0.06 |
| D | 5 | 3.76±0.77**# |
Table 1: EBD contents in lung tissues of each group.
Tie-2mRNA expression in whole blood
The relative expressions of Tie-2 mRNA in Groups D and B were significantly higher than those in groups A and C, among which the relative expression of Tie-2 mRNA in group D was nearly 5-fold of that in group B, and more than 50-fold than those in groups A and C, and the differences were statistically significant (Table 2, P<0.05).
| Group | Rats (n) | Tie-2 (2-??ct) |
|---|---|---|
| A | 6 | 0.32±0.11 |
| B | 6 | 3.99±0.93* |
| C | 6 | 0.60±0.16 |
| D | 6 | 19.72±2.01*# |
Table 2: Relative expressions of Tie-2 mRNA in whole blood in each group.
NO contents in serum and lung tissue homogenate
Group B exhibited the highest serum NO content, followed by group D, and there was significant difference between these two groups, the contents of NO in groups A and C were close, while far lower than those in groups B and D, and the differences were statistically significant (P<0.01, Table 3). The differences in the NO content in lung tissue homogenate among the groups were less than the differences in serum NO contents, group D exhibited the highest content, about 2-fold than group B, 3-fold than group C, and 5-fold than group A, there were statistically significant differences between group A and the other three groups (P<0.05), and the differences between group D and the other 3 groups were also statistically significant (P<0.05, Table 4).
| Group | Rats (n) | Serum NO content (μmol/l) |
|---|---|---|
| A | 6 | 22.66±3.18 |
| B | 5 | 188.30±5.18** |
| C | 5 | 31.57±8.14* |
| D | 5 | 123.13±4.24**# |
Table 3: Serum NO content in each group.
| Group | Rats (n) | NO content in lung tissues (μmol/l) |
|---|---|---|
| A | 6 | 10.37±1.29 |
| B | 6 | 23.63±3.92** |
| C | 6 | 16.94±1.62* |
| D | 6 | 53.62±6.70**# |
Table 4: NO content in lung tissue homogenate of each group.
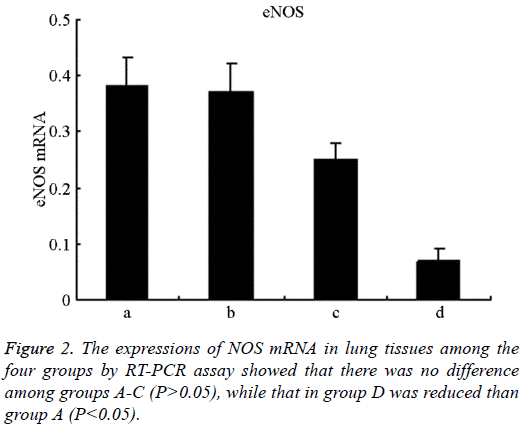
Expressions of NO synthase mRNA in lung tissues
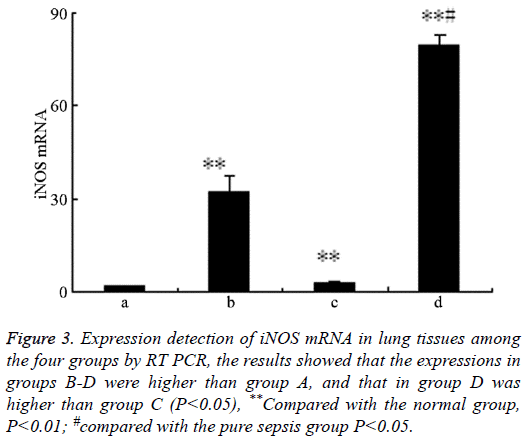
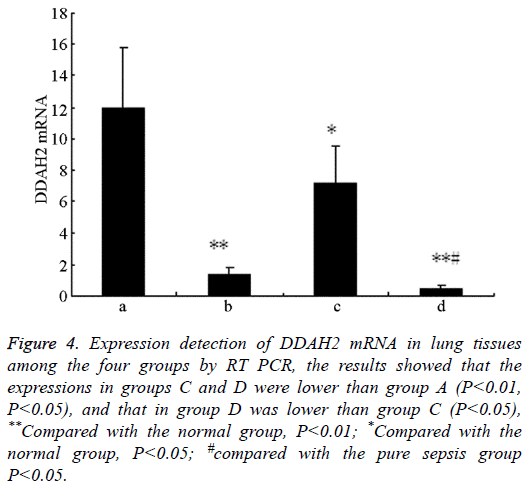
There was no difference in the expressions of eNOS among groups A-C, (P>0.05), while that in group D was lower than group A (P<0.05, Figure 2); the expressions of iNOS in groups B-D were higher than group A, and that in group D was higher than group C (P<0.05, Figure 3). The levels of DDAH2 mRNA in groups C and D were lower than group A (P<0.01 and P<0.05); and that in group D was lower than group C (P<0.05, Figure 4).
Figure 3: Expression detection of iNOS mRNA in lung tissues among the four groups by RT PCR, the results showed that the expressions in groups B-D were higher than group A, and that in group D was higher than group C (P<0.05), **Compared with the normal group, P<0.01; #compared with the pure sepsis group P<0.05.
Figure 4: Expression detection of DDAH2 mRNA in lung tissues among the four groups by RT PCR, the results showed that the expressions in groups C and D were lower than group A (P<0.01, P<0.05), and that in group D was lower than group C (P<0.05), **Compared with the normal group, P<0.01; *Compared with the normal group, P<0.05; #compared with the pure sepsis group P<0.05.
Discussion
Sepsis is a systemic inflammatory response caused by various pathogenic microorganisms or their toxins present inside blood or tissues. As the body's response to the infections, the status of the body would determine the degrees of its occurrence and severity. Normal body would not cause sepsis, and many diabetic patients were susceptible to the infection-caused sepsis, which is a common cause of death [13]. Numerous studies had shown that diabetes displayed the inhibitive status towards the inflammatory responses, namely the production of pro-inflammatory cytokines (TNF-α, IL-12, NO) would be reduced while the anti-inflammatory cytokines (IL-4, IL-10) [12-17] would be increasingly generated. These results seemed not to support the body’s excessive activation status when diabetic sepsis occurred, although diabetes or hyperglycemia exhibited the inhibitive status towards LPS-induced inflammatory responses, it might also be related with the fact that diabetes was prone to cause infections and clearance barriers towards the pathogens, which would resulted in the difficulties in controlling the infections, therefore inducing sepsis [18]. The pathological results of this study displayed that the inflammation changes in group D were not aggravated than group B, which also proved that the inflammatory responses were inhibited.
The occurrence of diabetes is based on microangiopathy pathologically, and the excessive activation of microvascular endothelial cells would be involved in the pathogenesis of sepsis, we speculated that when the infective factors stimulated, the endothelial cells in diabetic body might appear different changes, and were involved in the occurrence of sepsis. The indicators used for monitoring VECs functions included soluble mediators and circulating cells, the former included serum von Willebrand factor (vWF), soluble thrombomodulin, soluble adhesion molecules (E-selectin, intercellular adhesion molecule-1 and vascular cell adhesion molecule-1), and the latter included CEC and Endothelial Progenitor Cells (EPC). In contrast, as the indicator to assess the endothelial functions, thrombomodulin and adhesion molecules were susceptibly affected by other cells (platelets and neutrophils would also express these proteins), while vWF and circulating cells were more reliable. CEC is that inside the circulating blood, and under normal circumstances, certain number of CEC is the metabolic result of VECs. When in pathological situations, the number of CEC would exhibit can significant changes, therefore it could directly and specifically reflect the injury of CEC inside the body, and mainly reflect the shedding injury of endothelial cells [19]. Tie-2 is the indicator specific for endothelial cell injury [20], measuring the expression level of its mRNA could indirectly reflect the content of endothelial cells inside the circulation. This study replicated the diabetic rat model through intraperitoneally injecting STZ, when LPS induced sepsis, the much more serious lung microvascular permeability of EBD was observed than group B, the water content in lung and the endothelial cells inside the circulation were also increased, indicating the injury of endothelial cells was much more severe, so that the lung permeability of EBD was increased, the water content in lung was increased, suggesting the lung endothelial permeability was increased. The above results indicated that LPS could induce the diabetic patients to occur much more severe activation and injury in microvascular endothelial cells, as well as pulmonary edema, thus causing the occurrence of ARDS and sepsis.
Microcirculation includes capillaries, arterioles and venules, with the diameters ranging within 5-250 μm, the vast majority of the endothelial cells are distributed inside the microcirculation rather than the main arteries and veins. Therefore, microcirculation would be essential towards the homeostatic adjustment of cardiovascular functions and body organs’ functions. The activation and injury of endothelial cells is the main mechanism of microcirculation disorder, in which the compactness of endothelial cells are changed, endothelial cells shed from the basal membrane, thus exhibiting the increasing of vascular permeability and endothelial cells inside the circulation, leakage of macromolecules, migration and adhesion of leukocytes towards endothelial cells, followed by extensive tissue edema, injury and organ dysfunctions, even death [21]. Many mechanisms are involved in this pathophysiological process, among which the adjustment imbalance of iNOS and eNOS is one of the important pathogeneses [22]. Under physiological conditions, eNOS would be less expressed, with its important role to maintain the non-thrombotic surface of vascular endothelium. When in sepsis, its expression would be reduced [23], and its protective effects on vascular endothelial cells would thus disappear; meanwhile, it’s also an important molecule to maintain the tight junctions among vascular endothelial cells [24], which would particularly important for lung vascular functions. Studies had found that compared with the endothelial cells in the systemic circulation, the production of endothelial NO inside lung tissues was only protection factor towards LPS caused acute lung vascular injury [25], therefore, the generation of eNOS by NO would be particularly important for lung vascular functions. Bernardini found that LPS could decrease the activity and protein expression of eNOS in cultured bovine aortic endothelial cells [26]. 12 h culture of LPS could cause the expression reduction of eNOS in lung tissues [27]. And in sepsis, even the expression of eNOS was increased, it would be invalid, because decoupling would increase the NADPH oxidase-generated superoxide anion, so that the activity of NO would be decreased [28].
Furthermore, it might affect eNOS’s spatial conformation, making its distance to the cell membrane or Golgi membrane increased [29]. iNOS would be overproduced in sepsis and participate the injury towards the endothelial cells, while it would be hardly expressed under physiological conditions. Transgenic animal experiments had shown that the overexpression of eNOS could decrease the mortality in LPSinduced septic animals [30]; and the specific iNOS inhibitor LMAME could reduce the mortality. Several studies had demonstrated that the increasing of NO synthesis in lung tissues in ALI was caused by the increasing of iNOS induction [31-33].
Therefore, there existed NO adjustment imbalance in sepsis. This study confirmed there would be more serious NO adjustment imbalance in the patients with diabetes pus sepsis, which mainly manifested as: eNOS was reduced, the expression of iNOS was increased much more significantly, NO adjustment was much more disordered, so that the endothelial cell injury would be much more aggravated.
As for the NO levels, the contents of LPS were increased both inside the circulation and in lung tissues, while the local NO content in lung tissues would be further increased in diabetic sepsis, therefore it might be involved in the occurrence of lung microvascular injury [34]. The content in group B was lower, which might be interpreted as that the endothelial cells in large vessels would exhibit different reactions to LPS stimulation, thus participating the tension adjustment of large vessels. Studies had shown that after STZ induced diabetes, the intraperitoneal injection of LPS in diabetic rats exhibited no difference in the amount of NO produced by the aorta than that in normal rats after LPS injection, but the former exhibited the reduced reactivity towards phenylephrine than the latter [35].
In short, diabetes would aggravate the imbalance of endothelial NO system in LPS-caused sepsis, and it might be involved in endothelial cell injury, which manifested as the endothelial cell shedding inside the circulation and the pulmonary microvascular permeability were increased, tissue edema and organ dysfunction.
Acknowledgements
This study was supported by Special Joint Research Foundation of Doctoral Projects in colleges and universities (20102104110003).
Conflict of Interest
All authors have no conflict of interest regarding this paper.
References
- Zhou J, Xu H, Huang K. Organoselenium small molecules and chromium (iii) complexes for intervention in chronic low-grade inflammation and type 2 diabetesatural compound-derived epigenetic regulators targeting epigenetic readers, writers and erasers. Curr Top Med Chem 2016; 16: 823-834.
- Lindman BR, Goldstein JS, Nassif ME, Zajarias A, Novak E. Systemic inflammatory response syndrome after transcatheter or surgical aortic valve replacement. Heart 2015; 101: 537-545.
- Zechner D, Spitzner M, Müller-Graff T, Vollmar B. Diabetes increases pancreatitis induced systemic inflammation but has little effect on inflammation and cell death in the lung. Int J Exp Pathol 2014; 95: 411-417.
- Slynkova K, Mannino DM, Martin GS, Morehead RS, Doherty DE. The role of body mass index and diabetes in the development of acute organ failure and subsequent mortality in an observational cohort. Crit Care 2006; 10: 137.
- Vincent JL, Preiser JC, Sprung CL, Moreno R, Sakr Y. Insulin-treated diabetes is not associated with increased mortality in critically ill patients. Crit Care 2010; 14: 12.
- Ligtenberg JJ, Meijering S, Stienstra Y, van der Horst IC, Vogelzang M. Mean glucose level is not an independent risk factor for mortality in mixed ICU patients. Intensive Care Med 2006; 32: 435-438.
- Esper AM, Moss M, Martin GS. The effect of diabetes mellitus on organ dysfunction with sepsis: an epidemiological study. Crit Care 2009; 13: 18.
- Yang Y, Salam ZH, Ong BC, Yang KS. Respiratory dysfunction in patients with sepsis: protective effect of diabetes mellitus. Am J Crit Care 2011; 20: 41-47.
- Freebairn R, Park M. Systemic inflammatory response syndrome criteria for severe sepsis. N Engl J Med 2015; 373: 879-880.
- Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001; 29: 1303-1310.
- Khan FG, Ahmed E. Acute renal failure in diabetes mellitus. J Pak Med Assoc 2015; 65: 179-182.
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001; 25: 402-408.
- Schuetz P, Castro P, Shapiro NI. Diabetes and sepsis: preclinical findings and clinical relevance. Diabetes Care 2011; 34: 771-778.
- Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 2003; 348: 1546-1554.
- Hebbar K, Rigby MR, Felner EI, Easley KA, Fortenberry JD. Neuroendocrine dysfunction in pediatric critical illness. Pediatr Crit Care Med 2009; 10: 35-40.
- Klouche K, Da Mota EF, Durant R, Amigues L, Corne P, Jonquet O, Beraud JJ. Hypothalamic-pituitary-adrenal axis reactivity and dehydroepiandrosterone sulfate plasma concentrations in the critically ill elderly. Age Ageing 2007; 36: 686-689.
- Annane D. Adrenal insufficiency in sepsis. Curr Pharm Des 2008; 14: 1882-1886.
- Wijsman CA, Mooijaart SP, Westendorp RG, Maier AB. Responsiveness of the innate immune system and glucose concentrations in the oldest old. Age (Dordr) 2012; 34: 983-986.
- Takeuchi O, Sato S, Horiuchi T, Hoshino K, Takeda K. Cutting edge: role of Toll-like receptor 1 in mediating immune response to microbial lipoproteins. J Immunol 2002; 169: 10-14.
- Inohara, Chamaillard, McDonald C, Nunez G. NOD-LRR proteins: role in host-microbial interactions and inflammatory disease. Annu Rev Biochem 2005; 74: 355-383.
- Lavoie PM, Thibodeau J, Erard F, Sekaly RP. Understanding the mechanism of action of bacterial superantigens from a decade of research. Immunol Rev 1999; 168: 257-269.
- Shu X, Keller TC, Begandt D. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci 2015; 72: 4561-4575.
- Dinges MM, Schlievert PM. Role of T cells and gamma interferon during induction of hypersensitivity to lipopolysaccharide by toxic shock syndrome toxin1in mice. Infect Immun 2001; 69: 1256-1264.
- Sriskandan S, Altmann DM. The immunology of sepsis. J Pathol 2008; 214: 211-223.
- Li HY, Yao YM, Shi ZG, Dong N, Yu Y, Lu LR, Sheng ZY. The potential role of Staphylococcal enterotoxin B in rats with post burn Staphylococcus aureus sepsis. Shock 2003; 20: 257-263.
- Bernardini C, Greco F, Zannoni A, Bacci ML, Seren E, Forni M. Differential expression of nitric oxide synthases in porcine aortic endothelial cells during LPS-induced apoptosis. J Inflamm (Lond) 2012; 9: 47.
- Gross CM, Rafikov R, Kumar S, Aggarwal S, Ham PB, Meadows ML, Cherian-Shaw M, Kangath A, Sridhar S, Lucas R, Black SM. Endothelial nitric oxide synthase deficient mice are protected from lipopolysaccharide induced acute lung injury. PLoS One 2015; 10: 0119918.
- Rodiño-Janeiro BK, Paradela-Dobarro B, Raposeiras-Roubin S, Gonzalez-Peteiro M, Gonzalez-Juanatey JR, Alvarez E. Glycated human serum albumin induces NF- κB activation and endothelial nitric oxide synthase uncoupling in human umbilical vein endothelial cells. J Diabetes Complications 2015; 29: 984-992.
- Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med 2003; 348: 138-150.
- Remick DG. Pathophysiology of sepsis. Am J Pathol 2007; 170: 1435-1444.
- Hotchkiss RS, Tinsley KW, Swanson PE, Schmieg RE, Hui JJ, Chang KC, Osborne DF, Freeman BD, Cobb JP, Buchman TG, Karl IE. Sepsis induced apoptosis causes progressive profound depletion of B and CD4 T lymphocytes in humans. J Immunol 2001; 166: 6952-6963.
- Hack CE. Tissue factor pathway of coagulation in sepsis. Crit Care Med 2000; 28: 25-30.
- Nieuwland R, Berckmans RJ, McGregor S, Böing AN, Romijn FP. Cellular origin and procoagulant properties of microparticles in meningococcal sepsis. Blood 2000; 95: 930-935.
- Chuaiphichai S, Starr A, Nandi M, Channon KM, McNeill E. Endothelial cell tetrahydrobiopterin deficiency attenuates LPS-induced vascular dysfunction and hypotension. Vascul Pharmacol 2016; 77: 69-79.
- Taylor FB, Dahlback B, Chang AC, Lockhart MS, Hatanaka K. Role of free protein S and C4b binding protein in regulating the coagulant response to Escherichia coli. Blood 1995; 86: 2642-2652.