ISSN: 0970-938X (Print) | 0976-1683 (Electronic)
Biomedical Research
An International Journal of Medical Sciences
Research Article - Biomedical Research (2017) Volume 28, Issue 9
Anti-tumor activities of bamboo salt on sarcoma 180 tumor-bearing BALB/c mice
1Chongqing Collaborative Innovation Center for Functional Food, Chongqing University of Education, Chongqing, PR China
2Department of Food Science and Nutrition, Pusan National University, Busan, South Korea
3Department of Food Science and Biotechnology, Cha University, Gyeongghi-do, South Korea
#These authors have contributed equally to this work
- *Corresponding Author:
- Kun-Young Park
Department of Food Science and Biotechnology
Cha University, South Korea
Accepted date: February 3, 2017
Bamboo salt is a traditional Korean baked solar salt, processed by packing the solar salt (sea salt) in bamboo tubes and heating several times at high temperatures. The anti-tumor activity of bamboo salt was investigated and compared to those of other salt’s samples. Though solar salt and purified salt exhibited high tumor weight, bamboo salt is associated with a lower weight of tumor. Bamboo salt baked nine times (9X) showed greater increased in anti-tumor activity than salts baked once (1X) or three times (3X). The growth rate, of 9X baked bamboo salt in treatin mice Splenocytes was determined by a MTT Assay. This rate of proliferation was higher than that achieved with 1X baked bamboo salt by FBS, LPS or Con A. Purified and solar salts had relatively the lowest growth rate proliferation effects. Compared to the other salt samples, 9X bamboo salt significantly (p<0.05) reduced ALS, AST and BUN levels. The bamboo salts, especially 9X bamboo salt, also significantly (p<0.05) increased the cytokine levels of TNF-α and IL-1β compared to the other salt samples.
Keywords
Bamboo salt, Solar salt, Tumor, Sarcoma 180, BALB/c mice.
Introduction
Bamboo salt as a functional food, first produced in Korea, has been widely used in Easten Asia. Bamboo salt was first produced by a famous Buddhist Priest named; Jin-pyo Yul-sa, in Korea 1300 years ago [1]. For the manufacture process, the main raw materials of bamboo salt are solar salt (natural salt obtained from salt pond by sun drying) and bamboo grown for 3 years. Solar salt is put into a bamboo tube (about 8 cm in diameter and approximately 30 cm in length), that is sealed with natural red clay, and baked at 1000°C~1500°C using pine for fuel [2]. Bamboo salt baked once (1X) can be used as table salt. When the solar salt is baked in the bamboo case three (3X) or nine (9X) times, the salt absorbs minerals from the bamboo and clay and melts into a liquid during the baking process. The quality and medicinal properties of the bamboo salt are increased by more rounds (times) of baking. The 9X bamboo salt has been used as a traditional folk medicine in Korea [3]. Pharmaceutical research efforts have concentrated on evaluating the beneficial activities of bamboo salt such as; anti-cancer and anti-ulcer effects [4].
Bamboo salt is used as a folk medicine in Korea, after bamboo salt is baked more times; bamboo salt contains more iron, silicon, and potassium. These functional trace elements could make the bamboo salt have numerous pathologic effects, such as inflammation, viral diseases, diabetes and cancer [5].
The cancerous tumor is an abnormal growth of body tissues. In general, tumors occur when cells divide and grow excessively in the body. Normally, cell growth and division are strictly controlled [6]. Metastasis is the spreading of a cancer from, one organ or part to another non-adjacent organ or part. The new occurrences of the disease, thus generated, are referred to as metastases [7]. Metastasis is a multi-step process that begins when a primary tumor acquires mutations and becomes invasive, and cells eventually enter the blood or lymph [8]. Metastases arise most commonly in the; lungs, liver, brain, and bone. Interestingly, the lungs are the most common sites for systemic sarcoma metastases, because of the substantial vasculature that feeds into these organs in addition to particular tropic factors [9]. Other cancers that often metastasised to the lung include; breast, colon, prostate, bladder, and various Neuro-blastomas [10].
S180 cells are a kind of sarcoma cells, using these cells to build animal model could determine the anti-tissue tumor effects. The sarcoma 180 mouse cell line is derived, from a sarcoma carried in Swiss Webster mice and has been described to grow in multiple inbred mouse strains due to β2- microglobulin deficiency, MHC class I destabilisation, and lack of recognition by host CTLs. Injection of these cells results in strain-dependent killing due to accumulation of ascites fluid [11]. BALB/c mice are distributed globally and are among the most widely used inbred strains in animal experimentations. BALB/c mouse was often used for in vivo cancer researches. Sarcoma 180 tumor-bearing mice model was, a mellow research animal model, used for the tumor and metastasis studies [12].
Bamboo salt had an anti-colon cancer effect [5], bamboo salt also might has the anti-sarcoma effect, so in this study, we show that the bamboo salt anti-tumor effect in sarcoma 180- transplanted mice is used as a human tumor model. We used the model to compare the anti-tumor activities of: 1X, 3X, and 9X baked bamboo salts. The effects of these bamboo salts were compared with that of common salts (purified and solar salts). Additionally, serum levels and splenocyte cell proliferation were assessed.
Materials and Methods
Bamboo salt and other salts
Bamboo salt (1X, 1 time baked), bamboo salt (3X, 3 times baked), bamboo salt (9X, 9 times baked) and solar salt were provided by Taesung Food Company (Gochang, Jeonbuk, South Korea). Purified salt manufactured by Hanju Corporation (Ulsan, South Korea) was purchased in a local market (Jang Jeon-dong, Geum Jeong-gu, Busan, South Korea). These salts were added to the mouse diet to make the salt content diet 4.7%.
Animals
Male BALB/c mice (n=60, 6-week-old) were purchased from Samtaco (Osan, Gyeonggido, Korea, South Korea). They were maintained in a temperature controlled (temperature 25 ± 2°C, relative humidity 50 ± 5%) facility, with a 12 h, light/dark cycle and free access to a standard mouse diet and water.
Cell preparation
Mouse sarcoma 180 cells were purchased from the Korean Cell Line Bank of Seoul National University (Seoul, South Korea). A mouse was injected with sarcoma 180 cells, the sarcoma-180 cell line was cultured for 7-10 days in the abdominal cavity of a BALB/c mouse and the cultured cells were harvested with the peritoneal fluid and centrifuged at 1200 rpm for 10 min in Phosphate-Buffered Saline (PBS). The separated sarcoma cells were floated in PBS and centrifuged and their concentrations were adjusted to 1.0 × 106 cells/ml for the further animal experiment.
In vivo anti-tumor activity assay
The 0.2 ml of 1.0 × 106 cells/ml sarcoma 180 cells was implanted subcutaneously, in the left groins of control and sample group mice. The control and normal groups’ mice were fed with normal diet and water. The sample group mice were fed with purified, solar and bamboo salts (1X, 3X and 9X) diets and normal water for 28 days. Then, the tumors were removed and weighed [11]. The tumor-growth-inhibition ratio (I.R.) was calculated using the following formula: I.R. (%)=(Cw-Tw)/Cw × 100 (Cw: Average tumor weight of the control; Tw: Average tumor weight of the experimental group). The animal protocol used in this study was reviewed by the Animal Ethics Committee of Chongqing Medical University (SYXK (Yu) 2012-0001).
AST, ALT and BUN levels in serum
The mice plasma was collected and placed in room temperature for 1 h and then the plasma was centrifuged at 4500 rpm for 10 min to make serum. Aspartate Aminotransferase (AST), Alanine Aminotransferase (ALT) and (blood urea nitrogen) BUN levels of the serum were determined using serum determination available kits (Asan Pham, Seoul, South Korea).
Splenocyte proliferation assay
Splenocytes were obtained by gentle disruption of the spleen of BALB/c female mice and filtration via a 40 μm Nylon cell strainer (Falcon, NJ, USA). The erythrocytes were lysed with 0.38% NH4Cl-Tris buffer (pH 7.4), while the remaining cells were re-suspended in RPMI-1640 with 10 mM HEPES, 10% Fetal Bovine Serum (FBS), 100 mg/L streptomycin, and 100 IU/mL penicillin.
Splenocytes (1 × 106 cells/ml) were, respectively, treated with mitogens (lipopolysaccharide, LPS and concanavalinA, ConA) at 10 μg/ml, and co-cultured with test samples in 24-well plates for 24 h, 48 h and 72 h at 37˚C in a humidified atmosphere of 5% CO2. The proliferation of splenocytes was measured by an MTT Assay [13].
Analysis of inflammation-related cytokines in serum by ELISA
For serum cytokines Assay, blood from the inferior vena cava was collected into tube and centrifuged (3000 rpm, 10 min, 4˚C). The serum was aspirated and assayed as described below. Concentrations of inflammatory-related cytokines TNF-α and IL-1β (Bio-legend, San Diego, CA, USA) in serum were measured by ELISA according to the manufacturer’s protocol (Biolegend, CA, USA). Briefly, after biotinylated antibody reagent was added in 96-well plates, supernatants of homogenized serum were incubated at 37˚C in CO2 for 2 h. After washing with PBS, streptavidin-peroxidase (HRP) solution was added and the plate was incubated for 30 min at room temperature. The absorbance was measured at 450 nm using microplate reader [14].
Statistical analysis
An Analysis of Variance (ANOVA) was performed and the results are presented as the mean ± Standard Deviation (SD). Differences between mean values of the individual groups were assessed with a one-way ANOVA and Duncan's multiple range tests. Differences were considered significant when pvalues were<0.05. The SAS v9.1 statistical software package (SAS Institute Inc., Cary, NC, USA) was used to perform all statistical analyses.
Results
Tumor growth inhibitory effects
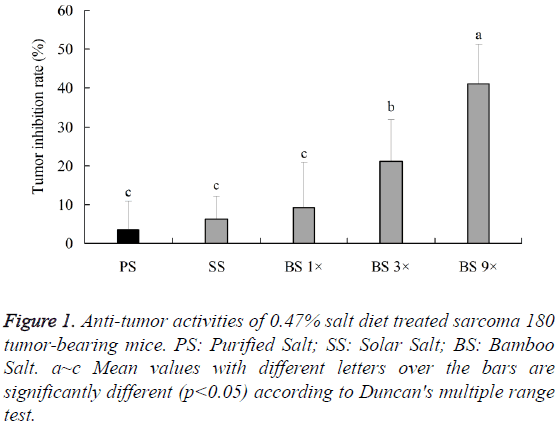
Bamboo salt 9X is the most famous bamboo salt in Korea, so we did the experiments for it. Anti-tumor activities of bamboo salts and common (purified and solar) salts were tested against sarcoma 180 tumor bearing mice (10 mice in each group). The average tumor weight of the sarcoma-180-cell-bearing mice after normal diet feeding (control) was 4.4 ± 0.6 g, whereas, that of the mice after purified and solar salts diet feeding was 4.2 ± 0.3 and 4.1 ± 0.6 g, and that of the mice after bamboo salt 1X, 3X and 9X diet feeding was 4.0 ± 0. 4, 3.4 ± 0.5 and 2.6 ± 0.5 g, were significantly (p<0.05) lower than the control group. According to the results, the bamboo salt 9X showed the best anti-tumor effect (41.2 ± 0.3%), bamboo salt 3X (21.2 ± 0.2%) and 1X (9.2 ± 0.2%) appeared better effects than the purified (3.6%) and solar salts (6.2 ± 0.1%) (Figure 1).
Effect of bamboo salt on serum levels of AST, ALT and BUN
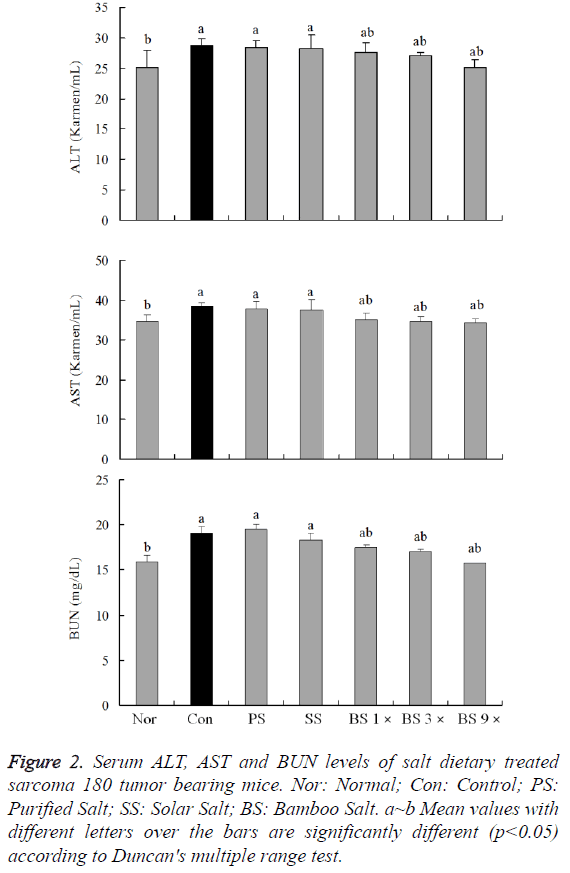
Serum levels of AST, ALT and BUN are the most important index for anti-sarcoma 180 tumor effects determination. The AST level of normal mice was 34.7 ± 1.7 Karmen/ml; however, that of sarcoma 180 control mice was significantly increased to 38.5 ± 1.0 Karmen/ml. The levels of AST in mice fed with purified salt, solar salt, bamboo salt 1X and 3X groups were 37.7 ± 2.0, 37.6 ± 2.6, 35.1 ± 1.7 and 34.7 ± 1.0 Karmen/ml, respectively. The levels of AST in rats treated with bamboo salt 9X decreased to 34.3 ± 1.0 Karmen/ml, which were lower than the levels in mice treated with other salts (Figure 2A).
Figure 2: Serum ALT, AST and BUN levels of salt dietary treated sarcoma 180 tumor bearing mice. Nor: Normal; Con: Control; PS: Purified Salt; SS: Solar Salt; BS: Bamboo Salt. a~b Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan's multiple range test.
The ALT level in the normal group was 25.1 ± 2.8 Karmen/ml, whereas that in the control group was 28.7 ± 1.2 Karmen/ml, reflecting a marked increase. The ALT levels in purified and solar salt groups decreased to 28.5 ± 1.1 and 28.2 ± 2.2 Karmen/ml, respectively. Bamboo salt 1X, 3X and 9X treatments resulted in even more decreased levels of ALT (27.6 ± 1.7, 26.9 ± 0.7 and 25.1 ± 1.3 Karmen/ml) (Figure 2B).
The levels of BUN in the purified and solar salt groups were 19.5 ± 0.7 and 18.3 ± 0.5 Karmen/ml, respectively, which were similar as the control group (19.1 ± 0.7 Karmen/ml). However, BUN level in the bamboo salt 1X and 3X groups was 17.5 ± 0.8 and 17.1 ± 0.3 mg/dL, bamboo salt 9X showed the lowest level of 15.8 ± 0.2 mg/dL, whereas the normal group showed the lower level of LDH, i.e. 15.9 ± 0.5 mg/dL (Figure 2C). The BUN levels in the bamboo salt groups were lower than that in the control group, similar to the findings of AST and ALT levels.
Effects of bamboo salt on lymphocytes proliferation
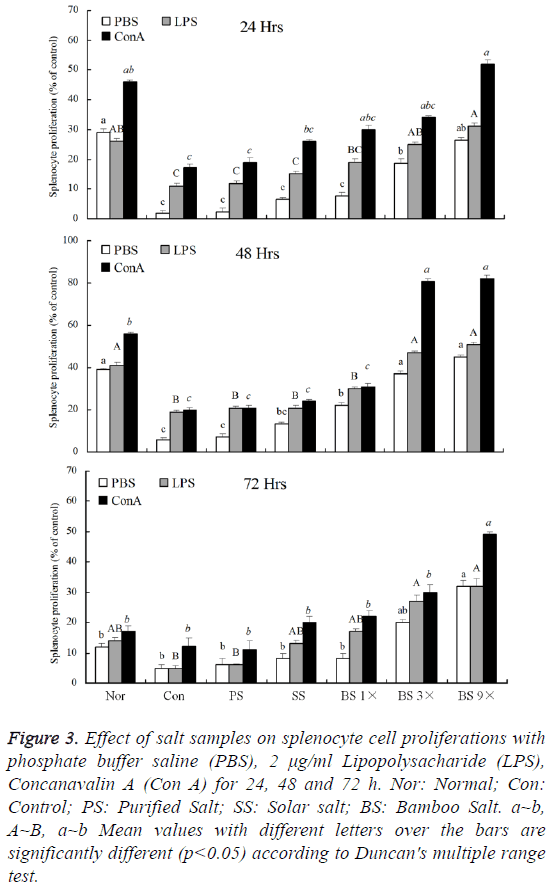
The lymphocytes of sarcoma 180 tumor bearing mice in each groups were irritated by LPS (lipopolysaccharide) or Con A (concanavalin A), these irritated lymphocytes were compared to the non-irritated PBS lymphocytes. As shown in Figure 3, after 24 h, the proliferation of Con A treated lymphocytes was more than LPS treated lymphocytes. Bamboo salt 9X group (52.2 ± 0.3%) showed the similar lymphocytes proliferation rate like normal group (46.1 ± 0.2%), and appeared the most lymphocytes proliferation in all salt groups. After cultured for 48 h, lymphocytes of all groups proliferated fast, Con A treated purified salt, solar salt, bamboo salt 1X, 3X and 9X group mice lymphocytes proliferation were 20.9 ± 0.2%, 24.1 ± 0.4%, 30.8 ± 0.3%, 81.0 ± 0.2% and 82.1 ± 0.3%, respectively. The lymphocytes were to continue culture for 24 h, the proliferation decreased compared to the 48 h. By these results, Con A treated lymphocytes proliferations of bamboo salt 9X fed mice were highest in all groups.
Figure 3: Effect of salt samples on splenocyte cell proliferations with phosphate buffer saline (PBS), 2 μg/ml Lipopolysacharide (LPS), Concanavalin A (Con A) for 24, 48 and 72 h. Nor: Normal; Con: Control; PS: Purified Salt; SS: Solar salt; BS: Bamboo Salt. a~b, A~B, a~b Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan's multiple range test.
Effect of bamboo salt on serum levels of TNF-α and IL-1β
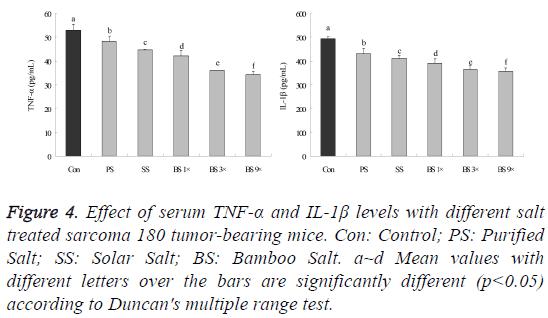
Serum TNF-α (tumor necrosis factor α) and IL-1β (interleukin-1β) levels in mice in the bamboo salt-treated groups (10 mice in each group) were significantly higher than those in the control (S180 treated) group (Figure 4, p<0.05). Decreased in TNF-α and IL-1β levels of bamboo salt 9X mice were 34.1 ± 1.7 and 358.5 ± 14.2 pg/ml, respectively, compared with the control (53.1 ± 2.5 and 493.2 ± 8.9 pg/ml) group (p<0.05). The TNF-α level in mice treated with purified salt, solar salt, bamboo salt 1X and bamboo salt 3X mice were 48.2 ± 2.3, 44.6 ± 0.2, 42.2 ± 2.1 and 36.1 ± 0.1 pg/ml, respectively. The IL-1β levels of those were 430.0 ± 22.3, 412.1 ± 10.9, 390.9 ± 18.4 and 364.9 ± 15.6 pg/ml, respectively.
Figure 4: Effect of serum TNF-α and IL-1β levels with different salt treated sarcoma 180 tumor-bearing mice. Con: Control; PS: Purified Salt; SS: Solar Salt; BS: Bamboo Salt. a~d Mean values with different letters over the bars are significantly different (p<0.05) according to Duncan's multiple range test.
Discussion
Bamboo salt has therapeutic effects on patients with various conditions, including: inflammation, viral diseases, diabetes, and cancer [2,4]. Evidence of the anticancer effects of the bamboo salts and underlying mechanisms were also observed in our previous experiments with HCT-116 and HT-29 colon carcinoma cells as well as AGS gastric carcinoma cells [5,15]. Those results demonstrated that the 9X bamboo salt has had the most potent in vitro anticancer effects, induced apoptosis, and anti-inflammatory activities. And bamboo salt also showed the in vitro anticancer and anti-inflammatory effect in oral cancer cell of TCA8113 human tongue carcinoma cells [16].
Cell injury can release endogenous damage associated with molecular patterns that, activate innate immunity. Cell injury can increase the risk of cancer [17]. AST and ALT are enzymes located in cells that leak out, into the general circulation when the cells are injured. AST is located in many body tissues, including the heart, muscle, kidney, brain and lung tissues [18]. A decreased BUN means creatinine ratio indicates malnutrition, and Syndrome of Inappropriate Antidiuretic Hormone secretion (SIADH), which is sometimes found with lung diseases, cancer, diseases of the central nervous system [19]. High levels of AST, ALT and BUN would in serum of tumor induced injury body.
Lymphocyte is a type of white blood cell in the vertebrate immune system, the three major types of lymphocyte are T cells, B cells and Natural Killer (NK) cells [20]. NK cells are a part of the innate immune system and play a major role in defending the host from both tumors and virally-infected cells. NK cells distinguish infected cells and tumors from normal and uninfected cells by recognizing changes of a surface molecule called MHC (Major Histocompatibility Complex). Class I NK cells are activated in response to a family of cytokines called interferon. Activated NK cells release cytotoxic (cell-killing) granules which then destroy the altered cells [21]. Lymphocyte decreasing would help the cancer development.
TNF-α and IL-1β are important regulators of host defense against tumor cells [22]. The observed increase in production of cytokines would suggest an enhanced ability of the host to combat the growth of tumors. Macrophages can be activated by β-glucans and other cell mediators to kill tumor cells by production of TNF-α and interleukins. The bioactivities of polysaccharides and polysaccharide-protein complexes are dependent on binding on the surface receptor of immune cells. These receptors, known as pattern recognition molecules, can recognize foreign ligands during the initial phases of the immune response [23]. At high levels of IL-1β, cancer cells receiving genotoxic insults, engage apoptotic pathways. Cotreatment with an inhibitor of IL-1β and TNF-α synthesis prevent carcinogen-induced lesions [24], the accumulation of IL-1 and TNF-α would lead to severe cancer.
Bamboo salts contain higher concentrations of minerals such as potassium, calcium, magnesium, and iron than purified or solar salts and the mineral contents of bamboo salts increase during the baking process [25]. Augmentation of the mineral contents is important for increasing anti-cancer activity [26]. Bamboo salt also contains more OH group than purified or solar salts, which may account for the fact that bamboo salt has a higher reduction potential [27]. Higher levels of minerals and more OH groups due to prolonged baking may have increased the anti-mutagenic, and anti-inflammatory and anti-tumor properties of the bamboo salt. The anticancer effects of the bamboo salts and underlying mechanisms were also determined in previous experiments with colon carcinoma cells as well as gastric carcinoma cells [5]. Those results showed that bamboo salt 9X had the best in vitro anticancer effect. Bamboo salt also showed the in vitro anticancer effects in TCA8113 human tongue carcinoma cells [16].
Conclusion
Results from the present study demonstrated that bamboo salts can decrease sarcoma 180 tumor-bearing tumor. The 9X baked bamboo salt has the greatest anti-tumor effects against MNNG compared to 1X or 3X bamboo salt as well as solar or purified salt. Bamboo salts also sarcoma 180 induced tumors. Bamboo salt, especially the 9X bamboo salt, has a strong activity as observed with tumor weight count, serum levels assay and lymphocytes proliferation. Increased mineral contents and reduction potential of the bamboo salts acquired during baking are important for augmenting these anti-tumor effects on sarcoma 180 treated tumors. Increasing the number of times, the bamboo salt was baked (up to nine times), particularly increased the properties of the salt in this study.
Acknowledgement
This work was supported by grant 20130290 to Solar Salt Research Center of Mokpo National University from Ministry of Oceans and Fisheries of Korea and the Program for Innovation Team Building at Institutions of Higher Education in Chongqing (grant no. CXTDX201601040).
References
- Shin HY, Lee EH, Kim CY, Shin TY, Kim SD, Song YS, Lee KN, Hong SH, Kim HM. Anti-inflammatory activity of Korean folk medicine purple bamboo salt. Immunopharmacol Immunotoxicol 2003; 25: 377-384.
- Yang JS, Seo J, Shin JH, Ahn YG, Lee DW. Polychlorinated dibenzo-p-dioxins and dibenzofurans from food salt samples processed by heat treatment in Korea. Chemosphere 2004; 54: 1451-1457.
- Shin HY, Na HJ, Moon PD, Shin T, Shin TY. Inhibition of mast cell-dependent immediate-type hypersensitivity reactions by purple bamboo salt. J Ethnopharmacol 2004; 91: 153-157.
- Kim HS, Kang SY, Jung KK, Kim TG, Han HM, Rheu HM, Moon AR. Characterization and anti-gastric ulcer activity of bamboo salt. J Fd Hyg Safety 1998; 13: 252-257.
- Zhao X, Kim SY, Park KY. Bamboo salt has in vitro anticancer activity in HCT-116 cells and exerts anti-metastatic effects in vivo. J Med Food 2013; 16: 9-19.
- Nowell PC. Mechanisms of tumor progression. Cancer Res 1986; 46: 2203-2207.
- Chiang AC, Massague J. Molecular basis of metastasis. N Engl J Med 2008; 359: 2814-2823.
- Wittekind C, Neid M. Cancer invasion and metastasis. Oncology 2005; 69 Suppl 1: 14-16.
- Vikis HG, Jackson EN, Krupnick AS, Franklin A, Gelman AE, Chen Q, Piwnica-Worms D, You M. Strain-specific susceptibility for pulmonary metastasis of sarcoma 180 cells in inbred mice. Cancer Res 2010; 70: 4859-4867.
- Songur N, Dinc M, Ozdilekcan C, Eke S, Ok U. Analysis of lung metastases in patients with primary extremity sarcoma. Sarcoma 2003; 7: 63-67.
- Tarnowski GS, Mountain IM, Stock CC. Influence of genotype of host on regression of solid and ascitic forms of sarcoma 180 and effect of chemotherapy on the solid form. Cancer Res 1973; 33: 1885-1888.
- Jung KO, Lee KY,Rhee SK, Park KY. Effects of various kinds of salt on the tumor formation, NK cell activity and lipid peroxidation in Sarcoma-180 cell transplanted mice. J Korean Assoc Cancer Prev 2002; 7: 134-142.
- Huang GC, Wu LS, Chen LG, Yang LL, Wang CC. Immuno-enhancement effects of Huang Qi Liu Yi Tang in a murine model of cyclophosphamide-induced leucopenia. J Ethnopharmacol 2007; 109: 229-235.
- Park HS, Park JY, Yu R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-alpha and IL-6. Diabetes Res Clin Pract 2005; 69: 29-35.
- Zhao X, Song JL, Lee JH, Kim SY, Park KY. Antioxidation and cancer cell (HT-29) antiproliferation effects of rubus coreanus Miquel bamboo salt. J Korean Assoc Cancer Prev 2010; 15: 306-312.
- Zhao X, Deng X, Park KY, Qiu L, Pang L. Purple bamboo salt has anticancer activity in TCA8113 cells in vitro and preventive effects on buccal mucosa cancer in mice in vivo. Exp Ther Med 2013; 5: 549-554.
- Seong SY, Matzinger P. Hydrophobicity: an ancient damage-associated molecular pattern that initiates innate immune responses. Nat Rev Immunol 2004; 4: 469-478.
- Delia R, Stefanovia M. Optimal laboratory panel for predicting preeclampsia. J Matern Fetal Neonatal Med 2010; 23: 96-102.
- Verbalis JG, Goldsmith SR, Greenberg A, Schrier RW, Sterns RH. Hyponatremia treatment guidelines 2007: Expert panel recommendations. Am J Med 2007; 120: 1-21.
- Caligiuri MA. Human natural killer cells. Blood 2008; 112: 461-469.
- Jangam SR, Yamada DH, McFall SM, Kelso DM. Rapid, point-of-care extraction of human immunodeficiency virus type 1 proviral DNA from whole blood for detection by real-time PCR. J Clin Microbiol 2009; 47: 2363-2368.
- Wells SM, Kew S, Yaqoob P, Wallace FA, Calder PC. Dietary glutamine enhances cytokine production by murine macrophages. Nutrition 1999; 15: 881-884.
- Gordon S. Pattern recognition receptors: doubling up for the innate immune response. Cell 2002; 111: 927-930.
- Roy D, Sarkar S, Felty Q. Levels of IL-1 beta control stimulatory/inhibitory growth of cancer cells. Front Biosci 2006; 11: 889-898.
- Zhao X, Kim SY, Park KY. Bamboo salt has in vitro anticancer activity in HCT-116 cells and exerts anti-metastatic effects in vivo. J Med Food 2013; 16: 9-19.
- Zhao X, Kim SH, Qi YC, Kim SY, Park KY. Effects of different kinds of salt in the comutagenicity and growth of cancer cells. J Korean Soc Food Sci Nutr 2012;41:26-32.
- Wilson HM, Chettibi S, Jobin C, Walbaum D, Rees AJ. Inhibition of macrophage nuclear factor-kappaB leads to a dominant anti-inflammatory phenotype that attenuates glomerular inflammation in vivo. Am J Pathol 2005; 167: 27-37.